
Newsroom
Malignant tumor treatment remains a challenge due to limited precision and significant side effects. Copper-based single-atom nanozymes have shown promise for tumor microenvironment-responsive precision therapy. However, their application is limited by weak substrate adsorption, difficulty in synthesizing low-coordination unsaturated structures, and issues brought up by preparation methods.
In a study published in Advanced Functional Materials, researchers from the Hefei Institutes of Physical Science of the Chinese Academy of Sciences (CAS), the University of Science and Technology of China of CAS, and the University of Macau, developed a coordination-unsaturated copper single-atom nanozyme.
Researchers proposed a ligand chelation conformation strategy. Using EDTA and copper chloride as precursors, they fabricated a carbon dot-supported, coordinatively unsaturated Cu-N2 single-atom nanozyme (Cu-N2-CDs) via a one-step hydrothermal method. The Cu-N2-CDs exhibited dual enzyme-like activities, mimicking both peroxidase and glutathione peroxidase in the tumor microenvironment.
Using the electron paramagnetic resonance spectrometer at the Steady High Magnetic Field Facility (SHMFF), researchers monitored the in situ generation of hydroxyl radicals (·OH), and revealed the structure-activity relationship between the coordination environment of the metal center and key factors such as substrate adsorption, electron transfer efficiency, and therapeutic performance.
Researchers also synthesized a coordinatively saturated Cu-N4 nanozyme (Cu-N4-CDs) for comparison. Experimental characterization and density functional theory calculations showed that compared with Cu-N4 sites, the unsaturated Cu-N2 configuration induced a high-spin, electron-rich state at the metal center.
The Cu-N2-CDs shifted the d-band center upward, enhanced H2O2 adsorption by 3.49 times, increased local electron density, and narrowed the band gap, thereby significantly accelerating electron transfer and ·OH generation, with a reaction rate up to 3.62 times higher than that of Cu-N4-CDs.
"Both in vitro and in vivo experiments showed that Cu-N2-CDs have strong anti-tumor effects. They significantly reduced cancer cell viability and performed better than Cu-N4-CDs in cell studies. In animal models, they also achieved much stronger tumor suppression. In addition, the nanozyme enables deep tumor imaging-guided therapy while maintaining good biosafety," said LIN Yefeng, one author of this study.
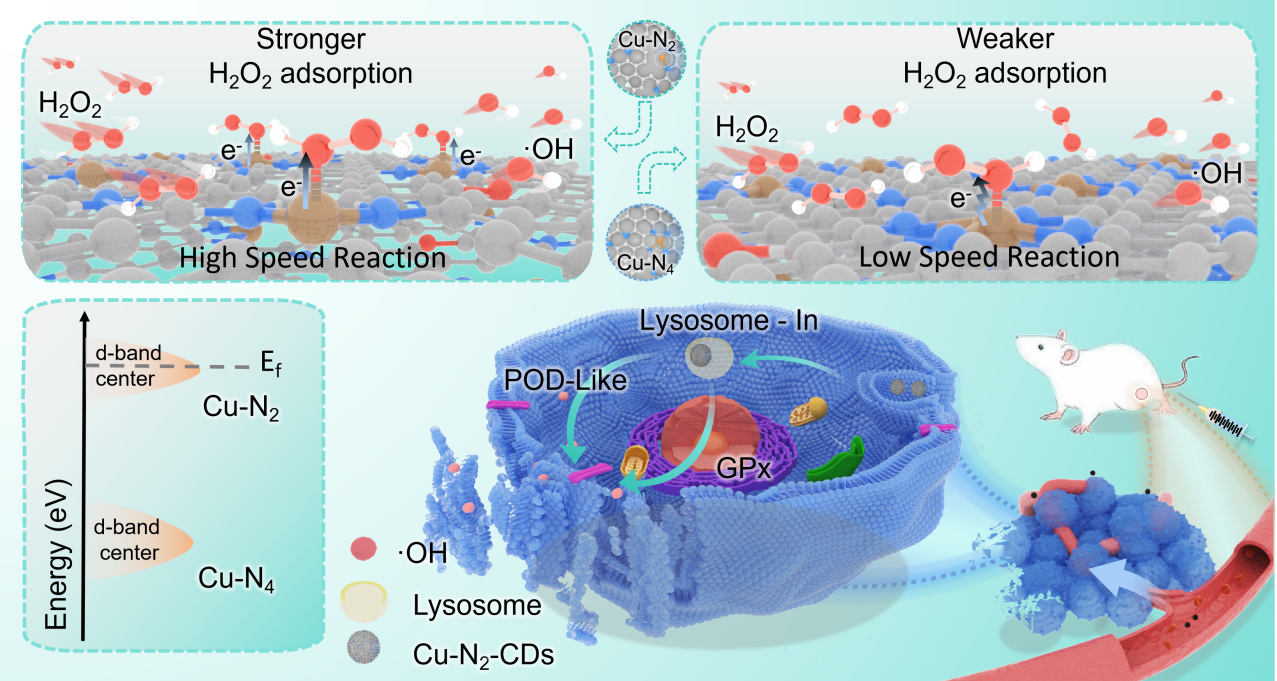
Schematic diagram of catalytic therapy mechanism of Cu-N2-CDs. (Image by LIN Yefeng)
