
Newsroom
Although mRNA vaccines have revolutionized medicine, generating strong, lasting T-cell immunity, which is critical for fighting cancer and persistent viruses, remains challenging. The core issue is delivery: current lipid nanoparticles (LNPs) act like a "blind drop," delivering mRNA indiscriminately to many cell types at the injection site, including non-immune cells. This inefficient targeting often results in weak cellular immune responses.
A research team led by Prof. XIA Yufei from the Institute of Process Engineering of the Chinese Academy of Sciences has developed a novel solution to this challenge by applying the chemical engineering concept of "process intensification" to vaccine design. They created a novel lipid nanoparticle-stabilized emulsion (LSE) system that shifts the focus from merely optimizing chemical formulas to actively controlling the physical delivery process.
The study was published in Cell Reports Medicine on March 17.
The LSE is not just another nanoparticle. It is a larger, structurally complex emulsion droplet (approximately 450 nm) designed to be preferentially taken up by the body's professional antigen-presenting cells (APCs), which are the key commanders of T-cell immunity. Think of it as changing the delivery vehicle and address to ensure the vaccine payload reaches the right "command center."
The results are striking. In a head-to-head test against the shingles, an LSE-delivered mRNA vaccine induced stronger T-cell responses that lasted over 100 days longer than those induced by the current clinical gold-standard vaccine, Shingrix®. In cancer models, it showed superior efficacy in preventing tumors and treating established cancers, resulting in long-term survival.
Mechanistic analyses further revealed that LSE intelligently redirects the immune response. While standard LNPs activate multiple, sometimes counterproductive, pathways, LSE preferentially channels the vaccine into the ideal pathway that efficiently educates killer T-cells, creating a potent and durable defense.
This work provides a powerful new platform. It moves mRNA vaccine technology from an era of "efficient expression" into an era of "precision immune programming", offering a versatile strategy for developing more effective vaccines against cancers and complex viral infections.
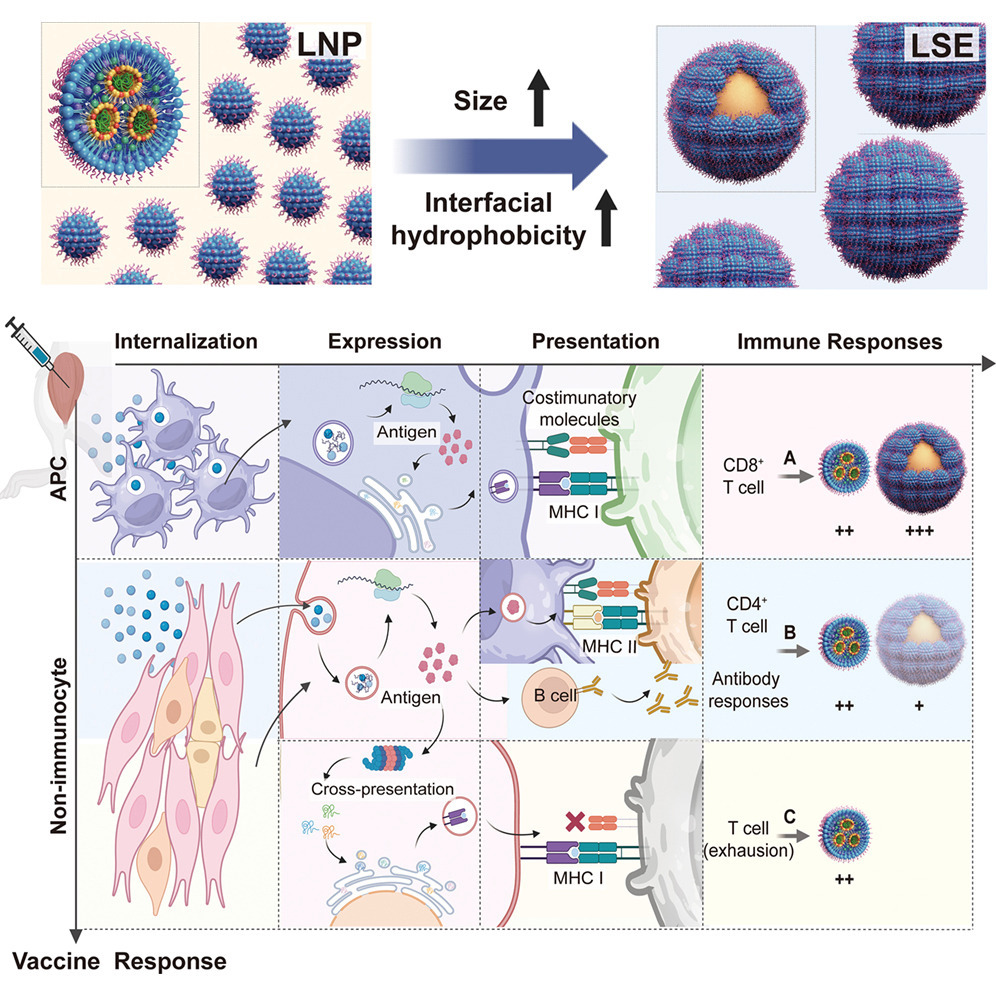
Lipid nanoparticle-stabilized Pickering emulsion (LSE) for APC-accentuated delivery and enhanced T cell immune responses (Image by GAO Weixiang)
