
Newsroom
Acute myeloid leukemia (AML) is one of the most aggressive hematologic malignancies, characterized by clonal expansion of undifferentiated myeloid progenitors. It predominantly affects the elderly population, and current treatment options remain largely limited to chemotherapy, with a five‑year survival rate of only about 30%. The emergence of chemotherapy‑resistant leukemia‑initiating cells (LICs) frequently leads to disease relapse, underscoring the urgent need for novel therapeutic targets that can effectively eliminate LICs without compromising normal hematopoiesis.
In a study published in Science Translational Medicine on May 6, a research team led by Dr. WANG Lan from the Shanghai Institute of Nutrition and Health (SINH) of the Chinese Academy of Sciences, along with the collaborators, uncovered a critical role of RNA binding protein with multiple splicing (RBPMS) in driving AML progression, and proposed a small‑molecule inhibitor targeting the RBPMS-FOXO1 axis.
The researchers found that RBPMS was upregulated in AML patients, and its high expression correlated with poor overall survival. Functional studies showed that RBPMS sustained the self‑renewal of leukemia stem cells and promoted leukemogenesis. In mouse models, genetic ablation of Rbpms had little effect on normal hematopoietic stem cell self‑renewal, multi‑lineage differentiation, or long‑term reconstitution capacity, suggesting that RBPMS represents an effective and safe therapeutic target.
Mechanistically, the researchers found that RBPMS recognized and bound specific motifs on FOXO1 mRNA through its RRM domain. It recruited the m6A reader IGF2BP3 to enhance FOXO1 mRNA stability in an m6A‑dependent manner, leading to the upregulation of FOXO1 protein. Moreover, the researchers found that RBPMS facilitated FOXO1‑mediated transcriptional activation of key glycolytic enzymes, thereby boosting glycolysis in AML cells.
Based on the structural information of the RBPMS RNA recognition motif domain, the researchers designed and screened a small‑molecule inhibitor. Using multiple models including AML mouse models, patient‑derived AML cells, and patient‑derived xenograft mouse models, they validated the therapeutic efficacy of the RBPMS inhibitor against AML, which holds significant implications and translational prospects for the clinical treatment of AML.
This study reveals the molecular mechanism by which RBPMS drives the progression of acute myeloid leukemia, establishes a regulatory link between RNA post-transcriptional regulation and metabolic reprogramming in AML, and provides a new strategy for precision therapy of AML.
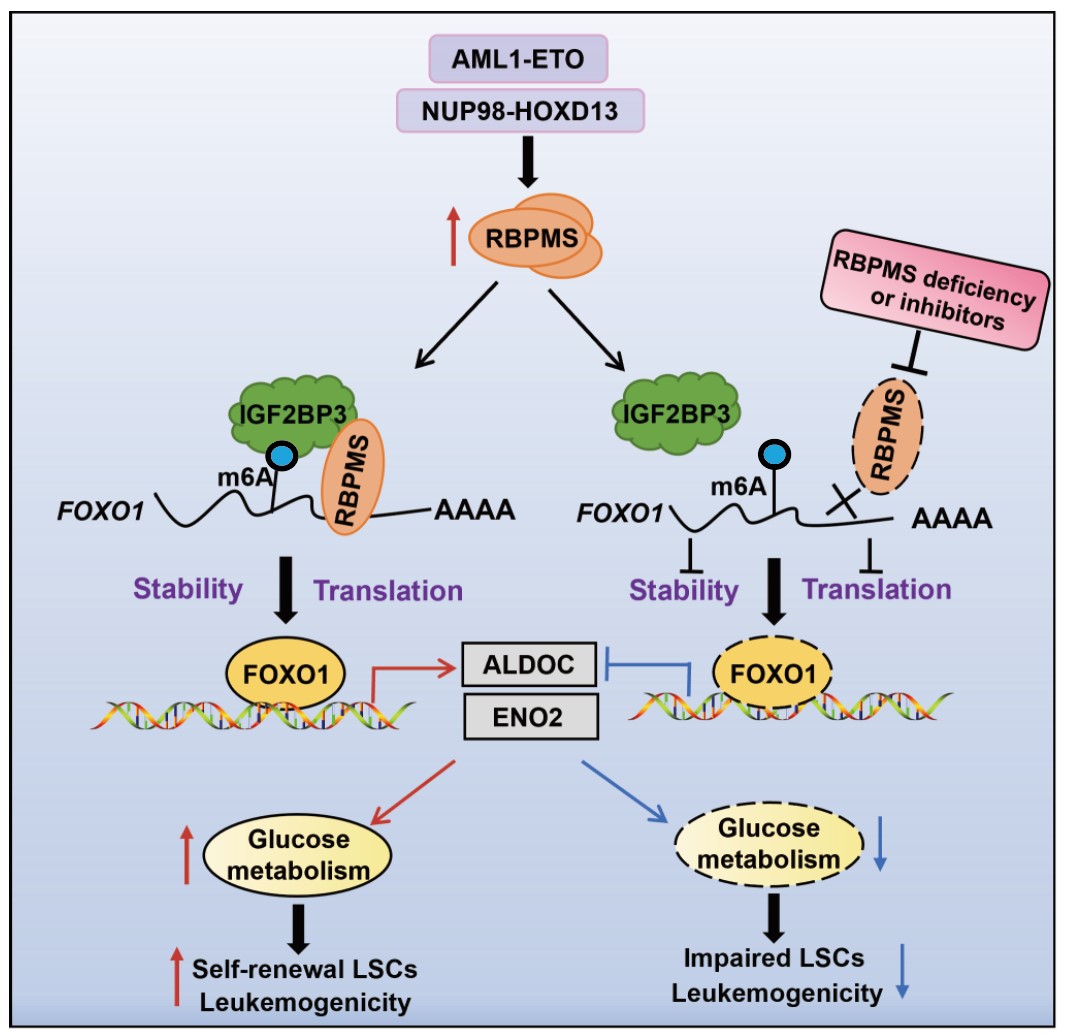
Molecular Mechanisms and Targeted Strategies for RBPMS Regulation of Acute Myeloid Leukemia Onset and Progression. (Image by Dr. WANG Lan's team)
