
Newsroom
A new study published in Cellon April 17 has unveiled an unexpected yet significant discovery: a previously unknown mechanism of membrane destruction termed "vertical membrane shearing." This discovery reshapes our understanding of bacterial immunity.
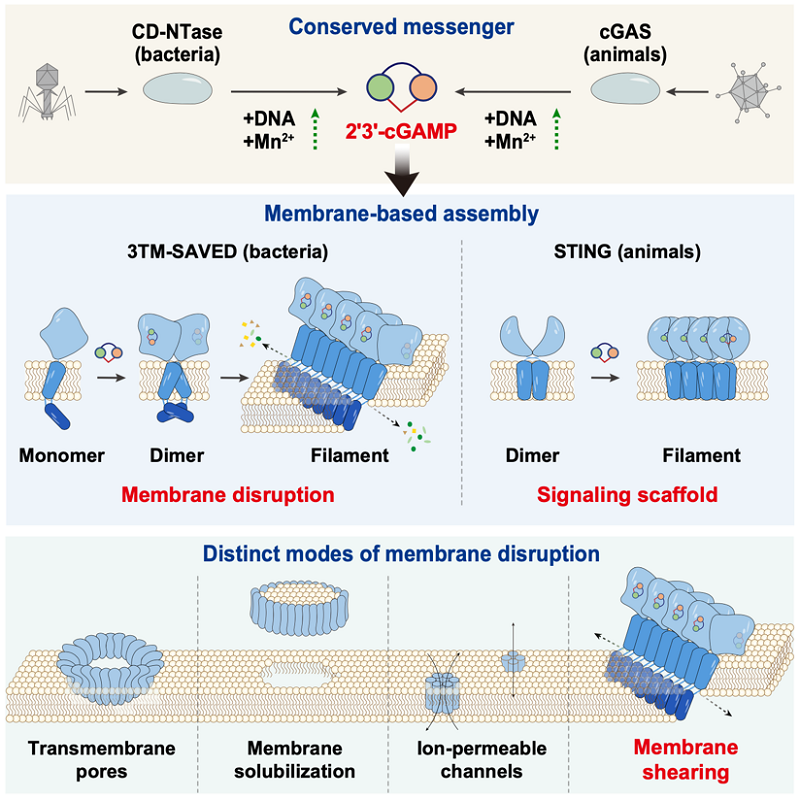
Led by Prof. GAO Pu from the Institute of Biophysics of the Chinese Academy of Sciences and Prof. GAO Ang from the Beijing Institute of Technology, the study systematically elucidates the production of 2'3'-cGAMP in prokaryotes, its activation of downstream transmembrane effector proteins, and how this signaling ultimately leads to a novel form of membrane damage and cell death.
The cGAS-cGAMP-STING pathway is a key DNA-sensing pathway in animal innate immunity, which transmits upstream DNA signals to the downstream membrane protein STING via the second messenger 2'3'-cGAMP and plays important roles in infection, inflammation, and tumor immunity. The unique 2'-5' and 3'-5' mixed phosphodiester bond structure of 2'3'-cGAMP confers stronger STING activation capability and profoundly influences the design of related agonists.
In this study, the researchers found that a class of bacterial CD-NTases is activated by DNA and Mn²⁺ and specifically synthesizes 2'3'-cGAMP. Further mass spectrometry and nuclear magnetic resonance analyses revealed that the chemical linkage of this product is identical to the 2'3'-cGAMP produced by mammalian cGAS, pointing to an evolutionarily ancient origen of this signaling logic.
They further focused on a representative and widespread class of transmembrane effectors-3TM-SAVED. Results showed that these proteins exist as monomers in the resting state. Upon specific recognition of 2'3'-cGAMP, they first form a transient dimeric intermediate and then assemble into higher-order filaments. Structural and functional analyses demonstrated that 2'3'-cGAMP directly participates in and stabilizes the protein assembly interfaces, driving the protein stepwise from a resting state to an activated state.
What happened next was unexpected. The 2'3'-cGAMP-induced higher-order filaments reorganize their own transmembrane helices and amphipathic hairpin motifs, allowing them to act together on the lipid bilayer in a mutually offset manner. This pulls the membrane into vertical misalignment, which the researchers call "vertical membrane shearing."
This shearing, the researchers revealed, is more than a structural deformation. It creates a linear array of small pores along the disturbed membrane interface. These pores are sufficiently large to allow the passage of water, ions, and small molecules, thereby significantly increasing membrane permeability, ultimately triggering cell death and blocking viral propagation.
This study expands our understanding of the evolutionary diversity of innate immunity across three levels: signaling molecules, receptor mechanisms, and effector execution. It broadens our knowledge of the evolutionary diversity of innate immune signaling systems and the mechanisms by which membrane proteins disrupt biological membranes.
2'3'-cGAMP-mediated diverse immune mechanisms (Image by GAO Pu's group)
