
Newsroom
Polarity is a physicochemical parameter of the cellular environment that has been shown to influence protein folding and aggregation in vitro. However, the polarity of different subcellular compartments has not yet been quantitatively compared, and its relationship with protein homeostasis is still largely unknown.
In a new study published in Communications Biology on February 3, researchers led by Prof. WANG Lei from the Institute of Biophysics of the Chinese Academy of Sciences have quantitatively mapped the polarity and crowding landscape of subcellular compartments and provides a new perspective for understanding and potentially intervening in protein conformational diseases.
They revealed that the endoplasmic reticulum (ER) has a distinctive environment, characterized by high polarity and molecular crowding. They also established a link between polarity and protein aggregation.
They used Halo proteins targeted to different subcellular compartments to localize the same polarity-sensitive probe to specific cellular regions. This strategy enabled the quantitative measurement and comparison of polarity in various subcellular environments in living cells.
Combined with a molecular crowding probe to assess crowding levels across compartments, the system established a technical foundation for dissecting the relationships among polarity, molecular crowding, and protein aggregation.
Using this approach, the researchers discovered that the lumen of the ER exhibits higher polarity and greater molecular crowding than the cytosol. This characteristic was consistently observed across multiple human cell lines.
Interestingly, the researchers found that the Huntingtin gene (Htt) exon1 with an expanded polyQ stretch (Htt-polyQ) has a lower tendency to form insoluble aggregates in the ER lumen (high polarity and greater crowding) than in the cytosol (low polarity and lesser crowding).
The researchers also observed that hypertonic treatments decrease polarity while increasing crowding. Under hypertonic treatment, the originally diffused Htt-polyQ in the cytosol further formed insoluble aggregates, indicating that polarity, rather than crowding, is correlated with protein aggregation.
This study reveals that the inherently high-polarity microenvironment of the ER may be a key factor in preventing protein aggregation. Together with the ER's specialized regulatory systems, this environment creates a robust protein quality control network that helps maintain proteostasis in cells.
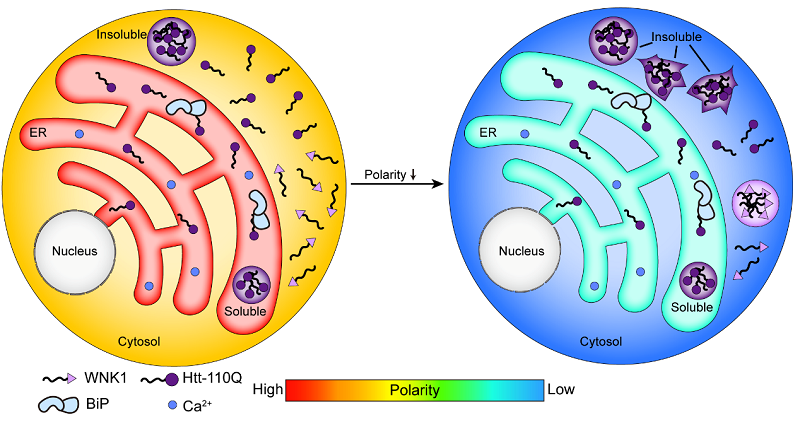
Schematic diagram of the model for polarity-regulated protein condensate and aggregate formation (Image by WANG Lei's group)
