
Newsroom
Endogenous viral elements (EVEs)—molecular fossils of ancient viral infections—are viral genetic fragments integrated into host genomes that can be transmitted across generations, offering a unique window into the evolutionary arms race between viruses and their hosts.
A research team led by Prof. LI Fuhua from the Institute of Oceanology of the Chinese Academy of Sciences (IOCAS), in collaboration with colleagues from Thailand's National Center for Genetic Engineering and Biotechnology (BIOTEC) and National Science and Technology Development Agency (NSTDA), has conducted a series of studies that shed new light on the dynamic process of viral endogenization in crustaceans.
The team's latest findings reveal the pervasive and dynamic integration of EVEs in crustacean genomes, including those derived from IHHNV and DIV1. The research provides critical insights into the long-term co-evolutionary arms race between crustaceans and their viral pathogens, while also highlighting urgent challenges for aquatic disease detection.
The researchers analyzed 105 crustacean genomes and identified 252 IHHNV-derived EVEs, which fall into two categories: ancient genomic integrations and rare modern insertions. These elements exhibit strict host specificity, being found exclusively in Decapoda, Thoracica, Isopoda, and Copepoda. Decapoda, in particular, showed the highest prevalence (82.9%) and a marked expansion of IHHNV-EVEs, reflecting an intensified evolutionary struggle with the pathogen. Viral non-structural protein 1 (NS1) accounted for 76.6% of all IHHNV-EVE integrations, linking the formation of these elements closely to viral replication processes.
Ancient IHHNV-EVEs were found to have ancestral inheritance traits. Collinear elements in giant isopods date back approximately 245 million years, while those in Penaeidae predate 100 million years. Additionally, three ancient EVEs are stably integrated in Litopenaeus vannamei across all wild and cultured populations. Only one authentic recent IHHNV integration was confirmed in Penaeus monodon, demonstrating that such insertions are ongoing but sporadic in crustaceans. Expression analyses showed that IHHNV-EVs are weakly expressed across L. vannamei tissues, with higher levels in gills, gonads, and the Oka lymphoid organ—key targets of IHHNV infection—suggesting potential vertical inheritance.
Additionally, the team uncovered a critical issue in crustacean disease detection: a high proportion of healthy L. vannamei (66.0%) and Macrobrachium rosenbergii (98.5%) tested positive for DIV1 using the WOAH-recommended ATPase-PCR assay, despite showing no pathognomonic DIV1 lesions or disease signs. While the PCR amplicons matched the pathogenic DIV1 sequence, bioassays using inoculums from these PCR-positive healthy decapods caused no mortality, viral replication, or pathological damage—unlike inoculums from diseased individuals.
Transmission electron microscopy confirmed the absence of intact DIV1 virions in healthy specimens. Metagenomic sequencing revealed only fragmented DIV1 sequences (602 mappable reads out of approximately 27 million), covering just 33% of the viral genome with no complete genomic sequence detected. In situ hybridization further distinguished these PCR-positive healthy samples, with signals confined to the nuclei of gill and hepatopancreas cells—unlike the cytoplasmic signals in target tissues observed in genuine DIV1 infections. These false-positive DIV1 diagnoses are likely caused by DIV1 viral inserts in host genomes acting as EVEs.
Complementary genomic analyses expanded these findings, leading to the establishment of a standardized bioinformatics pipeline for EVE identification and classification in crustaceans. The research also revealed universal yet lineage-specific viral endogenization across the 105 crustacean species studied. EVE integration was found to be a ubiquitous process in crustaceans but with distinct patterns across taxa: Decapoda primarily harbors EVEs from Nimaviridae and Retroviridae; Isopoda and Amphipoda from Adintoviridae; and Thoracica from a diverse range of viral families, including Nimaviridae, Parvoviridae, and Iridoviridae.
Notably, all EVE integrations strictly align with known host-pathogen relationships, with no cross-species viral insertions detected. Additionally, EVE flanking regions show lineage-specific enrichment of simple sequence repeats (SSRs) and transposable elements (TEs), confirming that EVE integration is not random but rather a genomic imprint of adaptive host-pathogen co-evolution.
Collectively, these studies redefine viral genomic colonization in crustaceans as a continuous, lineage-specific process, where the fixation of ancient EVEs coexists with ongoing modern integrations. EVEs emerge not only as molecular fossils of past host-pathogen conflicts but also as active participants in contemporary evolutionary battles, with some potentially conferring adaptive antiviral resistance.
The findings also carry practical implications: EVE-associated false PCR positives pose a threat to aquaculture trade, underscoring the need for revised disease detection protocols that combine genetic testing with histological and functional validation. For shrimp aquaculture, these insights lay the groundwork for EVE-based biomarkers and novel antiviral strategies, while deepening our understanding of the dynamic genomic co-evolution that has shaped crustacean-virus interactions over millions of years.
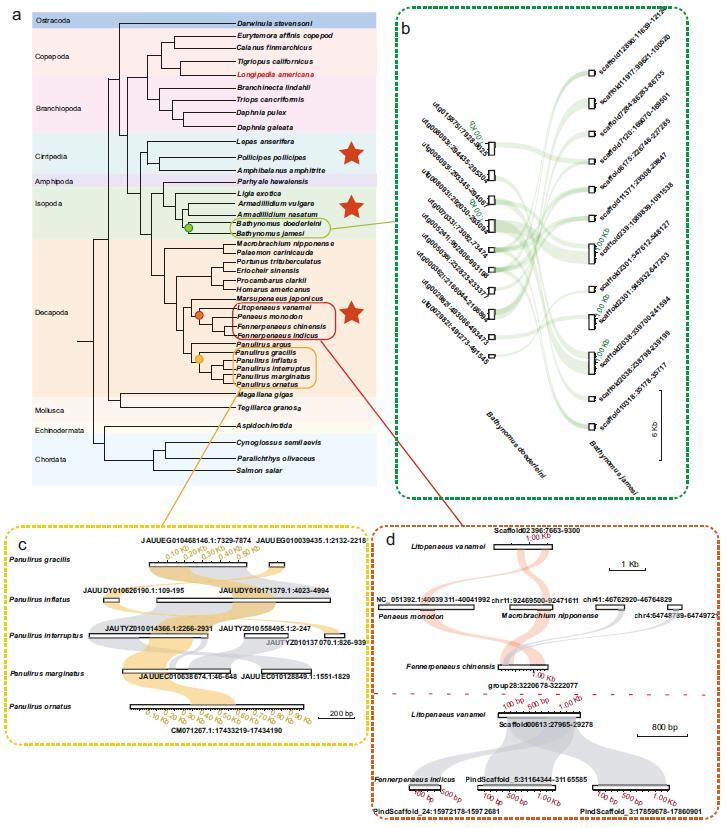
Integration of IHHNV in the crustacean genome. (Image by IOCAS)
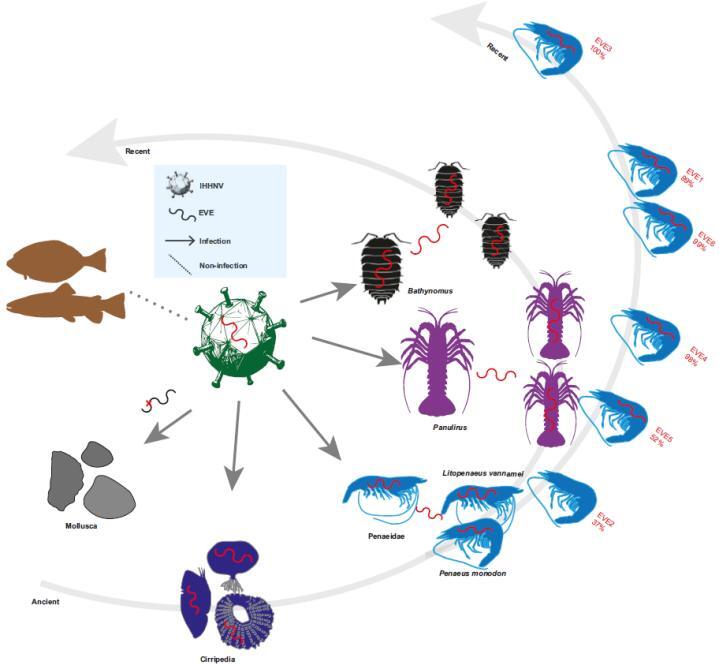
Dynamic integration model of IHHNV-EVEs in Crustaceans. (Image by IOCAS)
