
Newsroom
Scientists have developed a new pluripotent stem cell (PSC)-derived chimeric antigen receptor NK progenitor (CAR-NKP) cell therapy, providing a novel strategy for cancer immunotherapy.
CAR-NK cell therapy is regarded as safe and broadly applicable for the treatment of cancer and autoimmune diseases. However, CAR-NK cells show limited expansion and persistence in vivo, leading to suboptimal therapeutic efficacy. As a result, high cell doses and multiple infusions are often required to achieve durable clinical responses.
To address this challenge, a research team led by Prof. WANG Jinyong from the Institute of Zoology of the Chinese Academy of Sciences, has developed a CAR-PSC-derived NKP (CAR-iNKP) cell therapy that enables robust in vivo generation of long-lived CAR-iNK cells. The findings were published in Cell Stem Cell on February 24.
The team previously established an air-liquid interface organoid induction system for the large-scale production of induced NK (iNK) cells from human PSCs (hPSCs). Building on this work, the researchers identified a cell population phenotypically and functionally similar to natural NK progenitor (NKP) cells, which they named iNKP cells.
The hPSCs were engineered to express CXCR4, generating CXCR4-expressing iNKP (R4-iNKP) cells with improved bone marrow niche homing ability. A single low dose of R4-iNKP cells (2 × 105 cells per mouse) was injected into the tail veins of IL-15 humanized B-NDG mice. The cells successfully migrated to the bone marrow niche and differentiated into functional R4-iNK cells within seven to ten days. Notably, R4-iNK cells persisted in peripheral blood for more than 80 days. Furthermore, infusion of CAR-R4iNKP cells resulted in comparable long-term in vivo persistence of CAR-R4iNK cells.
The team evaluated the therapeutic potential of CD19- and CD7-targeted CAR-R4iNKP cells in human B-ALL and T-ALL animal models. Mice pre-infused with CAR-R4iNKP cells were fully protected against subsequent challenges with CD19- NALM6 and CD7- CCRF-CEM tumor cells, respectively.
When combined with chemotherapy, a single low-dose infusion of CAR-R4iNKP cells achieved complete long-term remission in B-ALL and reduced T-ALL relapse by 50%. In contrast, all control groups showed 100% tumor relapse. In addition, CAR-R4iNKP infusion alone significantly inhibited JeKo-1 lymphoma progression.
Cytokine-induced memory-like NK (MLNK) cells exhibit better in vivo persistence than conventional NK cells. Compared with PBMC-derived CAR-NK and CAR-MLNK cells, iPSC-derived CAR-R4iNK cells from the same donor displayed longer in vivo persistence. Importantly, only the CAR-R4iNKP group achieved long-term remission, while all mice treated with CAR-NK or CAR-MLNK experienced tumor relapse.
These findings demonstrate that CAR-iNKP cells overcome the limited persistence of conventional CAR-NK cells and show efficacy in eradicating tumor minimal residual disease (MRD). This study establishes a new strategy for tumor immunotherapy characterized by high efficacy, long-term persistence, and potential cost-effectiveness.
This research was supported by the National Natural Science Foundation of China and the Ministry of Science and Technology of the People's Republic of China.
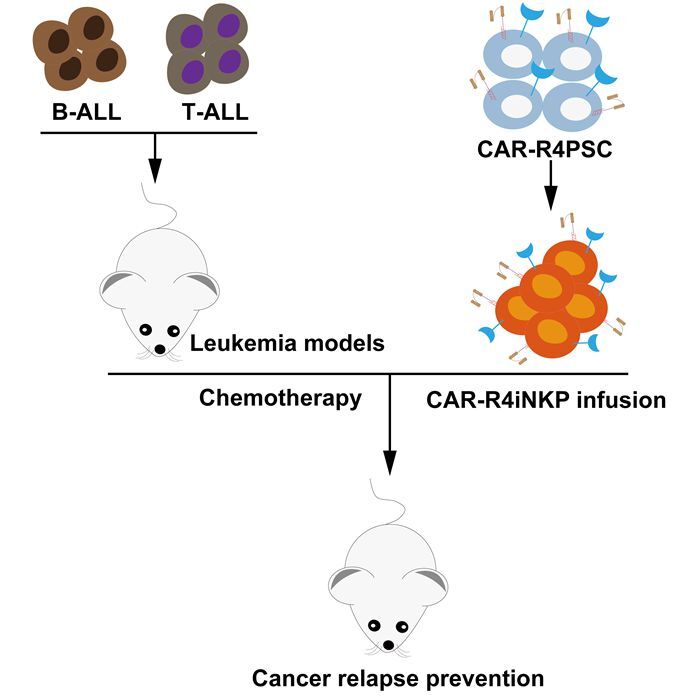
Schematic of CAR-iNKP Cell Therapy. (Image by WANG Jinyong's team)
