
Newsroom
A research team led by Prof. LIU Zhaoping at the Ningbo Institute of Materials Technology and Engineering (NIMTE) of the Chinese Academy of Sciences (CAS), in collaboration with researchers from the CAS Shenzhen Institutes of Advanced Technology and the University of Chicago, has systematically unraveled the lifespan failure mechanisms of ultrahigh-energy lithium metal batteries (LMBs).
The findings were recently published in Nature Reviews Chemistry.
Driven by the booming electric vehicle industry and the rapid development of the low-altitude economy, the development of battery technologies enabling ultra-long driving range and high-power output has become a core engine reshaping future transportation. A specific energy exceeding 600 Wh kg-1 is widely recognized as a critical milestone for next-generation battery systems.
Breaking through this energy threshold demands a dual-material approach: lithium-rich layered oxide cathodes that enable oxygen-redox reactions, and lithium metal anodes supporting reversible lithium plating and stripping. However, the researchers identified that this material combination induces complex electrochemical reactions and failure mechanisms distinct from those in commercial lithium-ion batteries (LIBs).
In contrast to commercial LIBs, where the cathode acts as the only lithium source, the team highlighted that LMBs exhibit a unique "dual-Li-reservoir" characteristic, with both the cathode and anode serving as lithium suppliers. This fundamentally changes the pathways of active lithium loss. During standard voltage-capacity tests, excess lithium in the anode continuously compensates for active lithium loss from the cathode, effectively masking the real depletion of active lithium. This frequently results in misinterpretations of capacity fade and the actual cycle life of the battery.
The study further clarified that macroscopic performance degradation stems from microscopic material failure. On the cathode side, deep anionic oxygen-redox leads to the collapse of transition-metal–oxygen polyhedra, triggering lattice instability and particle pulverization. On the anode side, the electrochemical lithium plating and stripping process promotes the formation of irreversible lithium dendrites and "dead lithium". These phenomena not only consume active lithium but also sharply increase cell impedance and induce cell swelling.
Reaching 600 Wh kg-1 requires extreme cell engineering, such as ultra-high areal cathode loadings and very low negative-to-positive (N/P) ratios. However, such extreme conditions come with trade-offs: they aggravate electrochemical inhomogeneity, accelerating local electrolyte depletion and material degradation.
The study provides a key insight: in practical applications, cell design and assembly processes often play a more important role in battery lifespan than material aging alone — a perspective frequently neglected in fundamental research.
This work opens the "electrochemical black box" of battery failure. It advocates for advanced in situ characterization techniques across multiple temporal and spatial scales to connect microscopic failure mechanisms with macroscopic practical performance, paving the way for the development of safer, longer-lasting, ultrahigh-energy-density batteries.
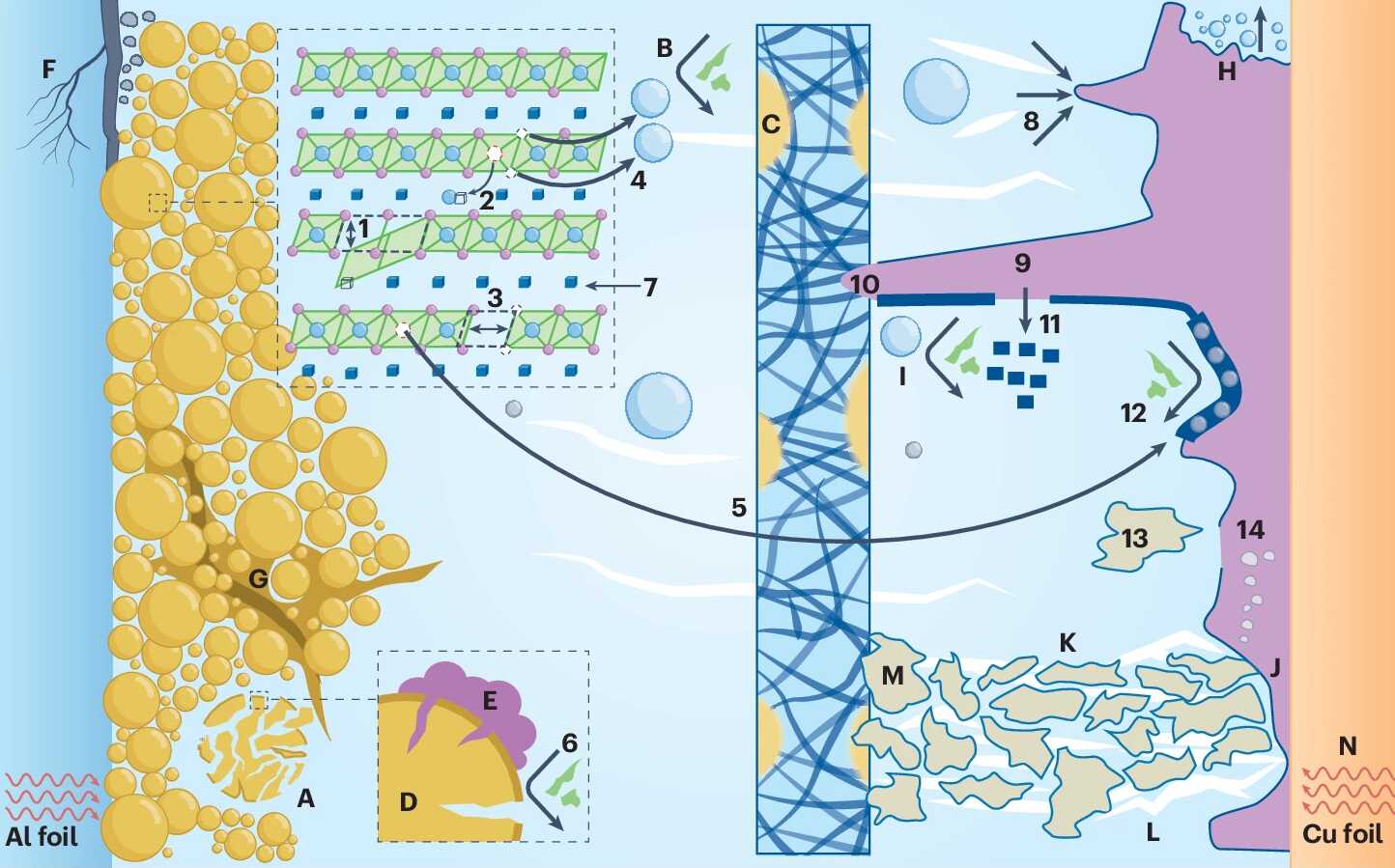
The lifespan failure mechanisms of lithium metal batteries. (Image by NIMTE)
