
Newsroom
Trained immunity confers innate immune memory through metabolic and epigenetic reprogramming, enabling innate immune cells to exhibit an enhanced immune response to secondary pathogenic stimulation. Although the epigenetic basis and functional changes underlying trained immunity in specific immune cell populations have been characterized, the intercellular communication mechanisms and master regulators that orchestrate multicellular immune coordination to establish system-wide trained immunity against infection remain incompletely understood.
Now, a research team led by Prof. LIU Cuihua from the Institute of Microbiology of the Chinese Academy of Sciences, in collaborating with Prof. PANG Yu from Beijing Chest Hospital, has identified a serum exosomal long non-coding RNA (lncRNA) associated with tuberculosis (TB) resistance. This lncRNA, termed TB Resister-derived CLOCK Regulator 1 (TRCR1), which drives intercellular immune training by modulating circadian regulator CLOCK-mediated epigenetic remodeling.
Their work was published in Cell Host & Microbe on December 30.
TB, caused by Mycobacterium tuberculosis (Mtb), has resurged as a leading global infectious disease killer. TB resisters are a special group of TB contacts who developed neither latent TB infection nor active TB despite prolonged and excessive exposure to Mtb. These individuals lack typical Mtb-specific adaptive immune signatures, such as T cell-mediated interferon-γ release, indicating that resisters may possess efficient innate immunity enabling early clearance of Mtb infection.
Previous work from Prof. LIU's group and collaborators demonstrated that monocyte-derived macrophages from TB resisters possess a stronger capacity to clear Mtb, leading to the hypothesis that their enhanced immune phenotype might be regulated by trained immunity.
In this new study, the researchers investigated serum exosomal lncRNAs from different Mtb-exposed populations via multi-omics analysis, identifying TRCR1 as a potent inducer of trained immunity.
Mechanistic experiments revealed that the Mtb antigen protein MPT53 stimulates lung epithelial cells to secrete exosomes carrying TRCR1, which are subsequently taken up by monocytes. In monocytes, TRCR1 collaborates with RNA-binding protein FXR2 to stabilize CLOCK mRNA by forming lncRNA-protein-mRNA complexes, thereby elevating CLOCK protein levels. Acting as a histone acetyltransferase, CLOCK then promotes histone H3 acetylation (K9/K14) at immune gene promoters, ultimately establishing epigenetic memory-mediated antimicrobial activity.
In mouse models, TRCR1 training strengthens anti-Mtb host immunity and improves Bacille Calmette-Guérin (BCG) vaccine efficacy.
Together, these findings reveal an intercellular immune training axis in which exosomal TRCR1 orchestrates CLOCK-mediated epigenetic programming to potentiate innate memory, providing a mechanistic framework and translational strategy to refine BCG vaccination and prevent infectious diseases.
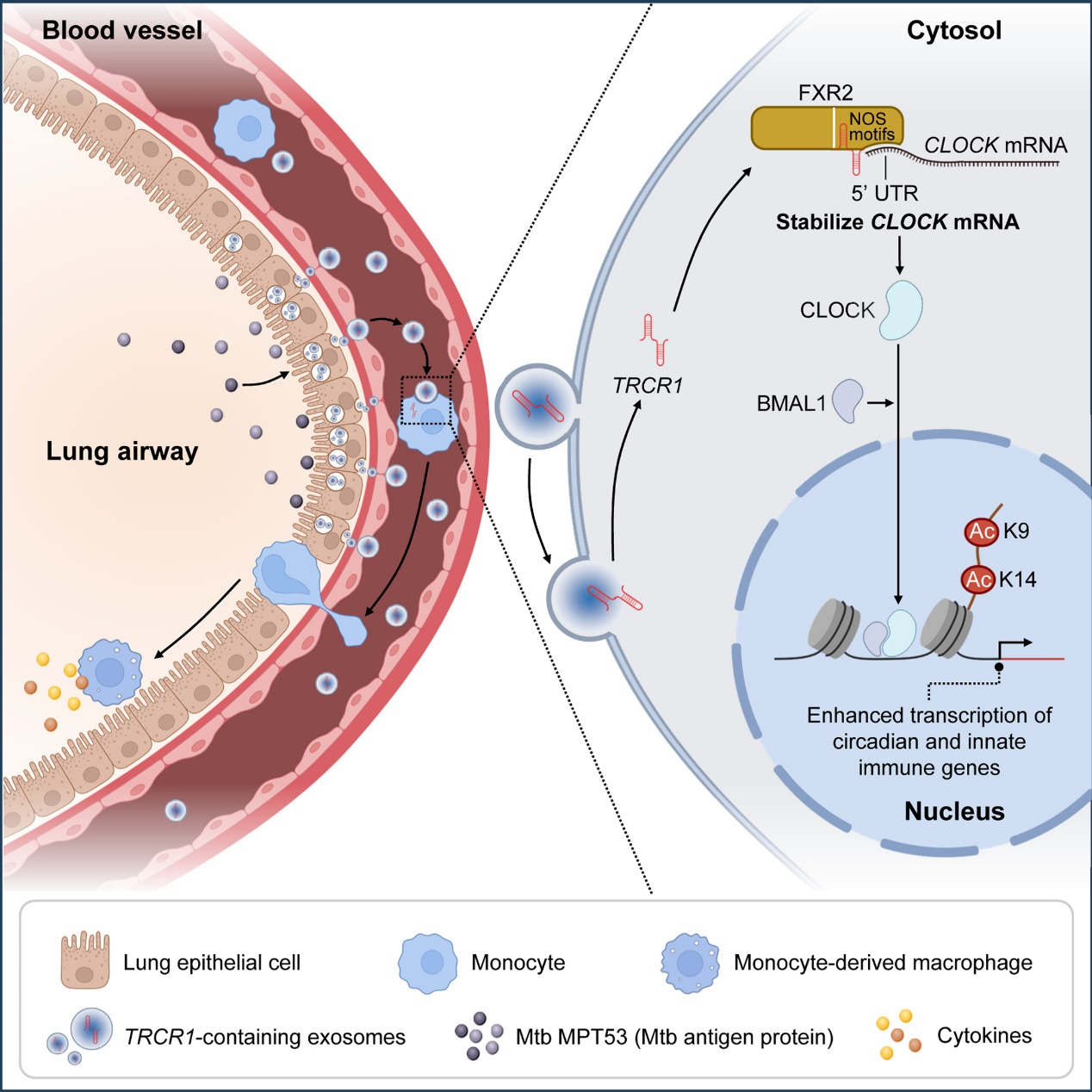
Exosomal lncRNA TRCR1 induces trained immunity by enhancing CLOCK-mediated epigenetic programming (Image by Prof. LIU Cuihua's group)
