
Mechanical sensations, including touch sensation, sound sensation, proprioception and mechanical nociception, are essential for animal survival. The molecular mechanisms underlying mechanical transduction are beginning to be elucidated. It has been known that mechanoreceptors respond to forces through mechano-gated ion channels since the responses are too rapid to involve second-messenger cascade. Recently, promising ion channel candidates, including only a few members of the Transient Receptor Potential (TRP), Degenerin/Epithelial Sodium Channel (DEG/ENaC) and Piezo families, have been identified.
31 members of the DEG/ENaC channel family have been identified in the Drosophila melanogaster genome to date. However, only Pickpocket (PPK) and Ripped Pocket (RPK) have been implicated in mechanosensation. To reveal novel DEG/ENaC channels that participate in mechanosensation, graduate students GUO Yanmeng and WANG Yuping from Dr. WANG Zuoren’s lab of the Institute of Neuroscience, Shanghai Institutes for Biological Sciences of Chinese Academy of Sciences, performed phylogenetic analysis of all the Drosophila DEG/ENaC channels and examined the expression pattern of several candidates.
Using the Gal4/UAS system, functional silencing and immunostaining studies, the researchers found that PPK26 are selectively expressed in class IV dendritic arborization (da) neurons. The class IV da neurons, which detects intense mechanical forces and harmful heat as well as intense short-wave light, represents the polymodal nociceptor in Drosophila larvae. Previous work showed that PPK, Pain, and Piezo proteins all contributed to mechanical nociception in the class IV da neurons. Here, the researchers found that PPK26 contributes to mechanical nociception but not thermal nociception in class IV da neurons, functioning together with PPK.
Genetic interactions studies indicate that PPK26 and PPK function in the same pathway in mechanical nociception while Piezo functions in a parallel pathway. The researchers found that PPK and PPK26 are interdependent with each other for their plasma membrane localization, while Piezo does not affect their plasma membrane localization. This work provides new clues for future studies on mechanical nociception in mammals.
This study entitled “The Role of PPK26 in Drosophila Larval Mechanical Nociception” was published on Cell Reports on November 06, 2014.
The work was supported by the Strategic Priority Research Program of the Chinese Academy of Sciences and China 973 Project.
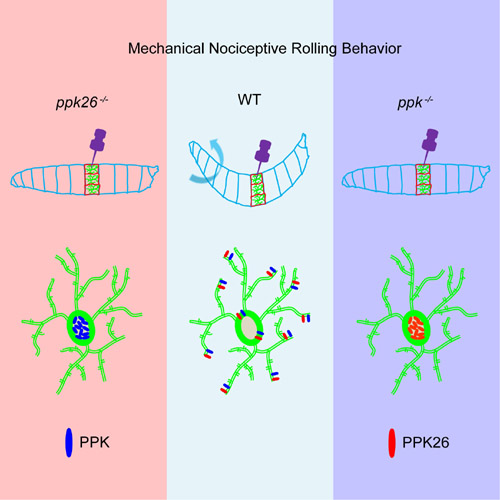
Left: ppk26 mutant larvae exhibit remarkably reduced rolling behavior in response to mechanical noxious stimuli and PPK could not translocate to the plasma membrane. Middle: Wild type larvae respond to mechanical noxious stimuli with rolling behavior. Right: ppk mutant larvae exhibit remarkably reduced rolling behavior in response to mechanical noxious stimuli and PPK26 could not translocate to the plasma membrane. (Image by Dr. WANG Zuoren)

86-10-68597521 (day)
86-10-68597289 (night)

52 Sanlihe Rd., Xicheng District,
Beijing, China (100864)

