
Both FJSM-GAS-1 and FJSM-GAS-2 were found to exhibit really rapid ion exchange kinetics, that is, they could complete the ion exchange within five minutes and 15 minutes, respectively.
Moreover, researchers revealed that both FJSM-GAS-1 and FJSM-GAS-2 could maintain the ion exchange ability and excellent selectivity for [UO2]2+ ion, even in the pH range of 2.9-10.5 and with the existence of Na+, Ca2+ and HCO3- in large quantity. The KdU of FJSM-GAS-1 could reach 6.06 × 10 6 mL/g, which is the highest value among that of the reported U adsorbents.
They showed that the [UO2]2+-laden products of FJSM-GAS-1 and FJSM-GAS-2 could be eluted in a simple and inexpensive way to recover uranium.
The afore-mentioned advantages, combined with the easy synthesis and excellent radiation resistances of β and γ irradiation make FJSM-GAS-1 and FJSM-GAS-2 promising candidates as the materials for capturing radioactive [UO2]2+, Cs+ and Sr2+ from nuclear waste solutions.
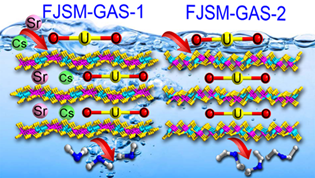
Schematic presentation of the [UO2]2+ , Cs+ and Sr2+ ion exchanges by FJSM-GAS-1 and FJSM-GAS-2 (Image by Prof. HUANG’s Group)

86-10-68597521 (day)
86-10-68597289 (night)

86-10-68511095 (day)
86-10-68512458 (night)

cas_en@cas.cn

52 Sanlihe Rd., Xicheng District,
Beijing, China (100864)

