
Flexible metal-organic frameworks (FMOFs) not only have permanent porosity and structural diversity, but also can respond to the external stimuli such as heat, sound and light while keeping their crystallinity. Due to the transformations between narrow pores to large pores (or between non-porous structure and porous structure), these FMOFs show potential applications in the storage and separation of gases.
In a study published in Angew. Chem. In. Ed., the research group led by Prof. HONG Maochun from Fujian Institute of Research on the Structure of Matter (FJIRSM) of the Chinese Academy of Sciences (CAS) reported a series of robust FMOFs. These flexible materials have high stability and can keep their integrity under acidic condition.
Compared with the robust MOFs, FMOFs have very high bromine uptakes up to 4.2 g·g-1. Additionally, more than 80 percent of bromine will remain in FMOFs after 24 hours while almost all of liquid bromine is gasified within one hour.
Single crystal X-ray experiments revealed that bromine molecules can be encapsulated into the pores of the MOFs. At the same time, the crystallographic axes of these FMOFs expand with the coefficients up to 4.5% after bromine adsorption.
The above results indicated that the flexibility of the FMOFs allows for structural adaptation upon bromine uptake to afford stronger host-guest interactions and therefore higher bromine adsorption capacities.
In addition, bromination reactions using binaphthol as the substrate indicated that the efficiency of bromine-pre-absorbed FMOFs is much higher than that of the rigid MOF or liquid bromine under ambient conditions.
The study revealed excellent repeatability in bromine capture and release capacities with bromination yields of 75% and 62% after three cycles of adsorption-bromination tests, respectively. This indicated that these FMOFs show promise in practical applications.
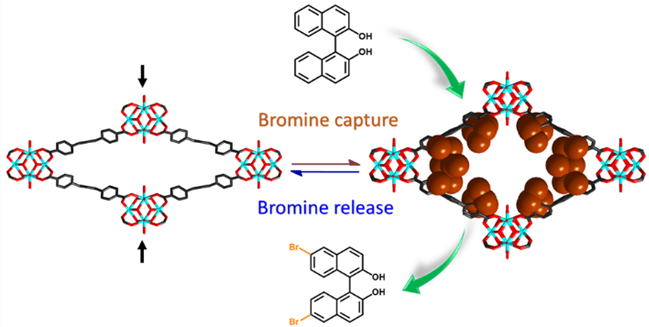
Schematic representation of the reversible bromine capture and release by the FMOFs. (Image by Prof. HONG’s Group)

86-10-68597521 (day)
86-10-68597289 (night)

86-10-68511095 (day)
86-10-68512458 (night)

cas_en@cas.cn

52 Sanlihe Rd., Xicheng District,
Beijing, China (100864)

