
Due to the unique properties of fluorine atom(s), fluorinated compounds play important roles in agrochemicals, pharmaceuticals, and materials science. Over the past decade, impressive achievements have been efficiently made in the introduction of fluorinated group into organic molecules.
However, most of these approaches used well-known are expensive fluorinating reagents, the use of inexpensive, abundant and widely available industrial raw material fluoroalkanes have received less attention because of their relatively inert reactivities.
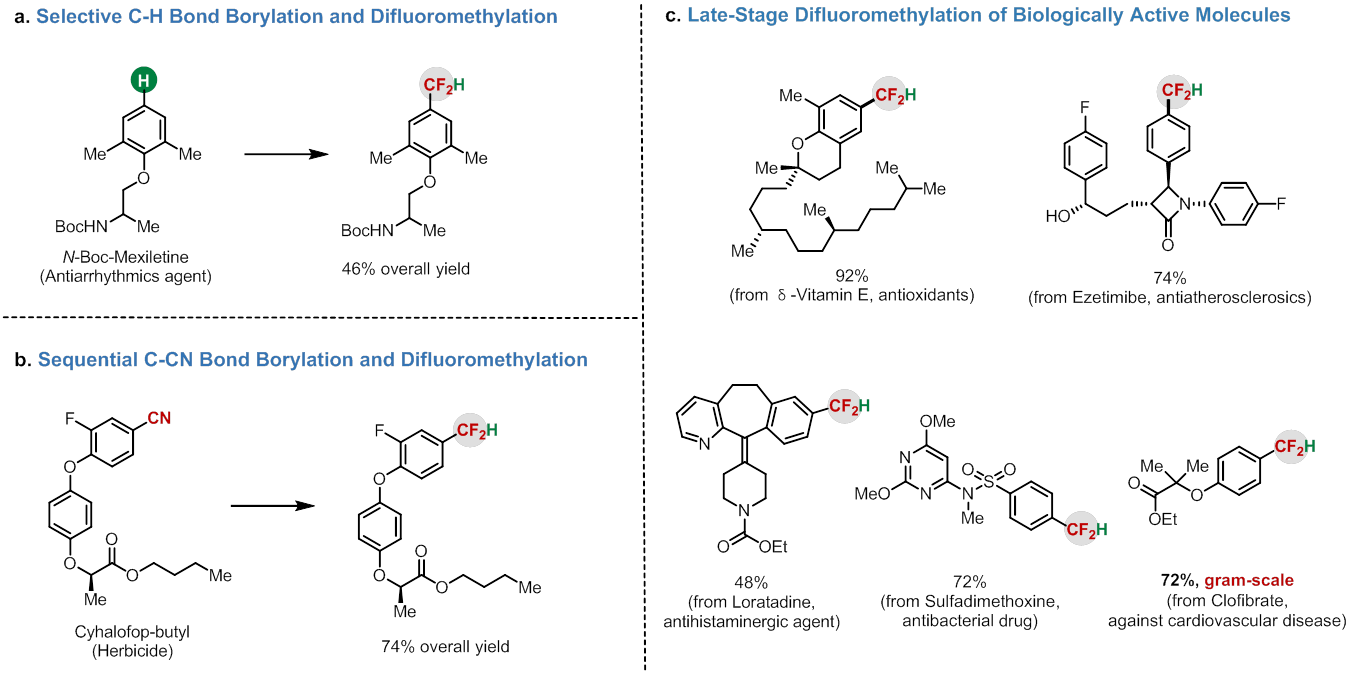
Most recently, ZHANG Xingang's group from Key Laboratory of Organofluorine Chemistry, Shanghai Institute of Organic Chemistry, Chinese Academy of Sciences, reported the first example of direct formation of difluoromethylated arenes from inexpensive and widely available industrial raw material ClCF2H.
CF2H is not only considered as a bioisostere of a hydroxy and thiol group, but also functions as a lipophilic hydrogen bond donor. The selective introduction of CF2H onto aromatic rings can significantly improve their bioactivities compared with their non-fluorinated counterparts.
Thus, difluoromethylation is a useful strategy for the modification of biologically active compounds. However, the difluoromethylating reagents used in previous work are expensive and require multistep synthesis.
ClCF2H (R22) is the most inexpensive and abundant industrial raw material used for the production of various fluorinated polymers (i.e. polytetrafluoroethylene, PTFE) (Figure 1a).
From the point view of cost-efficiency and step-economy, ClCF2H would be an ideal and straightforward difluoromethylating reagent, however, the use of ClCF2H for difluoromethylation of aromatics remains a challenge and has not been reported.

Figure 1. Activation of ClCF2H in organic synthesis. (Image by ZHANG Xingang)

86-10-68597521 (day)
86-10-68597289 (night)

86-10-68511095 (day)
86-10-68512458 (night)

cas_en@cas.cn

52 Sanlihe Rd., Xicheng District,
Beijing, China (100864)

