
Membrane-membrane protein interaction is one of the research hotspots of biology science. The components and structures of cell membranes are quite complicated. Some lipids directly interact with some membrane proteins and mediate their activities. For example, cholesterol and phosphatidylinositol-4,5-bisphosphate (PIP2) can directly interact with G protein-coupled receptors (GPCRs) and voltage-gated potassium (Kv) channels, respectively.
Crystallography studies have revealed several cholesterol binding sites on some GPCRs. Molecular dynamics (MD) simulations have directly observed the binding of cholesterol molecules to several GPCRs, obtaining results consistent with the crystal structures on the known cholesterol binding sites, and suggesting several new binding sites. However, the interactions and influences of PIP2 on Kv channels remain elusive. The function of KCNQ2 channel, an epilepsy-associated Kv channel, is dependent on PIP2 lipids in the membranes. Recently, researchers from JIANG Hualiang’s group and the International Scientist Working Station of Neuropharmacology supervised by LI Min, Shanghai Institute of Materia Medica (SIMM), provide insight into the mechanism of PIP2 action on KCNQ channels.
Previous studies on the Shaker and Kv1.2 channels have shown that PIP2 decreases the voltage sensitivity of Kv channels. Interactions between PIP2 and the S4 segment or the S4-S5 linker in the closed state have been highlighted to explain the effects. Using MD simulations, the researchers of SIMM showed that PIP2 preferentially interacts with the S4-S5 linker in the open state KCNQ2 channel, whereas it contacts the S2-S3 loop in the closed state. These interactions are different from the PIP2-Shaker and PIP2-Kv1.2 interactions. Consistently, PIP2 exerts different effects on KCNQ2 relative to the Shaker and Kv1.2 channels, i.e., PIP2 upregulates the voltage sensitivity of the KCNQ2 channel.
The Shaker, Kv1.2 and KCNQ2 channels have similar structural arrangements and voltage sensing mechanisms. The differential interactions of PIP2 with these channels contribute to the diversity of PIP2 regulations. This study suggests that the effects of lipids and their interactions with membrane proteins are critical determinants for the diverse functions of highly homologous membrane proteins. Membrane-membrane protein interactions should be studied at a finer scale.
On November 25, this study was released in PNAS.
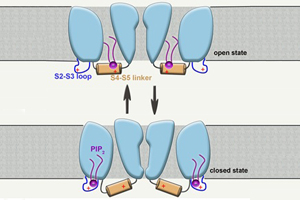
Mechanism of PIP2 action on the KCNQ2 channel (Image by SIMM)

86-10-68597521 (day)
86-10-68597289 (night)

86-10-68511095 (day)
86-10-68512458 (night)

cas_en@cas.cn

52 Sanlihe Rd., Xicheng District,
Beijing, China (100864)

